Alkenes and Alkynes
Part I: Properties and Synthesis
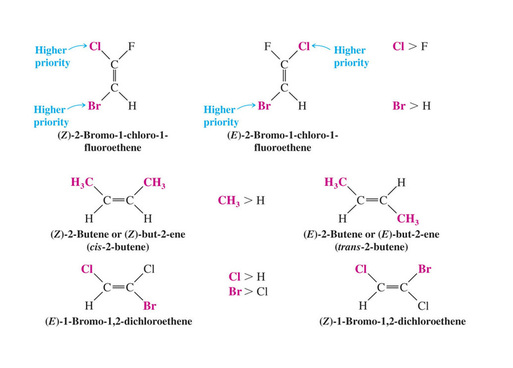
The (E)-(Z) System for Designating Alkene Diastereomers
The Cahn-Ingold-Prelog convention is used to assign the groups of highest priority on each carbon. Higher priority is given to atoms with higher atomic number. If they are the same element, look at the next atom attached to it.
If the group of highest priority on one carbon is on the same side as the group of highest priority on the other carbon the double bond is Z (zusammen)
If the highest priority groups are on opposite sides the alkene is E (entgegen)
The Cahn-Ingold-Prelog convention is used to assign the groups of highest priority on each carbon. Higher priority is given to atoms with higher atomic number. If they are the same element, look at the next atom attached to it.
If the group of highest priority on one carbon is on the same side as the group of highest priority on the other carbon the double bond is Z (zusammen)
If the highest priority groups are on opposite sides the alkene is E (entgegen)
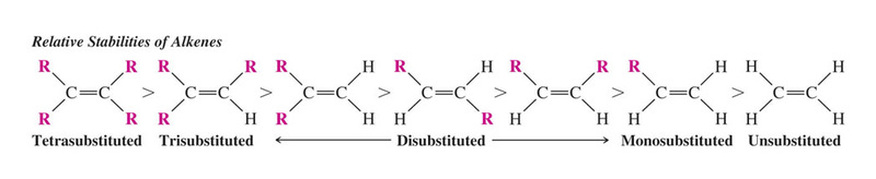
Relative Stabilities of Alkenes
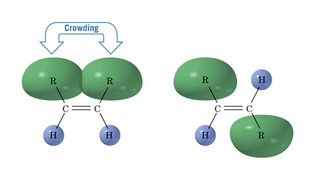
Generally cis alkenes are less stable than trans alkenes because of steric hinderance
Generally cis alkenes are less stable than trans alkenes because of steric hinderance
Overall Relative Stabilities of Alkenes
The greater the number of attached alkyl groups (i.e. the more highly substituted the carbon atoms of the double bond), the greater the alkene’s stability
The greater the number of attached alkyl groups (i.e. the more highly substituted the carbon atoms of the double bond), the greater the alkene’s stability
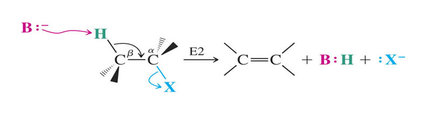
Synthesis of Alkenes via Elimination reactions
*Dehydrohalogenation
- Reactions by an E2 mechanism are most useful
E1 reactions can be problematic
E2 reaction are favored by:
Secondary or tertiary alkyl halides
Alkoxide bases such as sodium ethoxide or potassium tert-butoxide
Bulky bases such as potassium tert-butoxide should be used for E2 reactions of primary alkyl halides
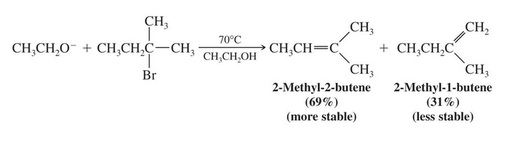
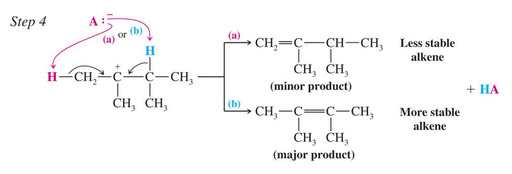
Zaitsev’s Rule: Formation of the Most Substituted Alkene is Favored with a Small Base
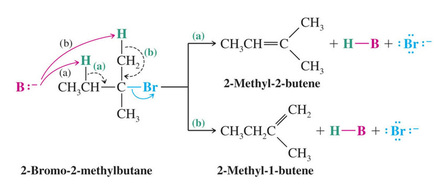
Some hydrogen halides can eliminate to give two different alkene products
Some hydrogen halides can eliminate to give two different alkene products
Zaitzev’s Rule: when two different alkene products are possible in an elimination, the most highly substituted (most stable) alkene will be the major product. This is true only if a small base such as ethoxide is used.
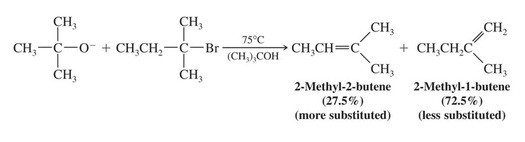
Formation of the Least Substituted Alkene Using a Bulky Base
Bulky bases such as potassium tert-butoxide have difficulty removing sterically hindered hydrogens and generally only react with more accessible hydrogens (e.g. primary hydrogens).
Bulky bases such as potassium tert-butoxide have difficulty removing sterically hindered hydrogens and generally only react with more accessible hydrogens (e.g. primary hydrogens).
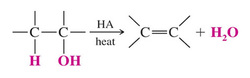
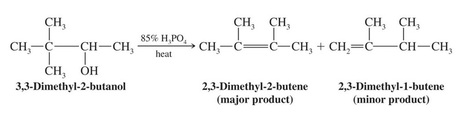
*Acid - Catalyzed Dehydration of Alcohols
>Recall that elimination is favored over substitution at higher temperatures
>Typical acids used in dehydration are sulfuric acid and phosphoric acid
>The temperature and concentration of acid required to dehydrate depends on the structure of the alcohol
>Primary alcohols are most difficult to dehydrate, tertiary are the easiest.
>Typical acids used in dehydration are sulfuric acid and phosphoric acid
>The temperature and concentration of acid required to dehydrate depends on the structure of the alcohol
>Primary alcohols are most difficult to dehydrate, tertiary are the easiest.
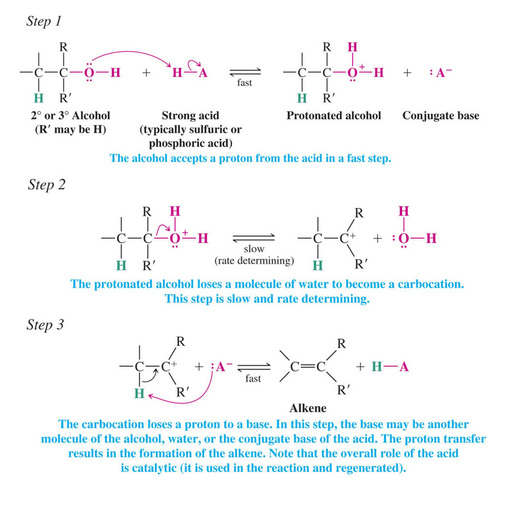
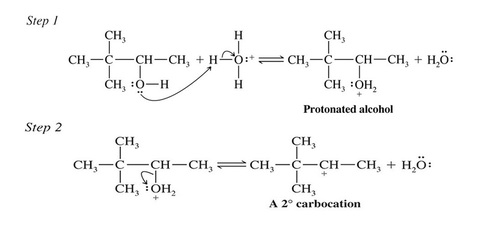
Mechanism for Dehydration of Secondary and Tertiary Alcohols: An E1 Reaction
Only a catalytic amount of acid is required since it is regenerated in the final step of the reaction
Only a catalytic amount of acid is required since it is regenerated in the final step of the reaction
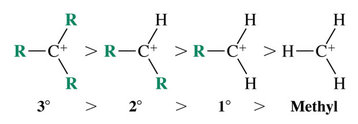
Carbocation Stability and the Transition State
Recall the stability of carbocations is:
Recall the stability of carbocations is:
>>The second step of the E1 mechanism in which the carbocation forms is rate determining
>>The transition state for this reaction has carbocation character
>>Tertiary alcohols react the fastest because they have the most stable tertiary carbocation-like transition state in the second step
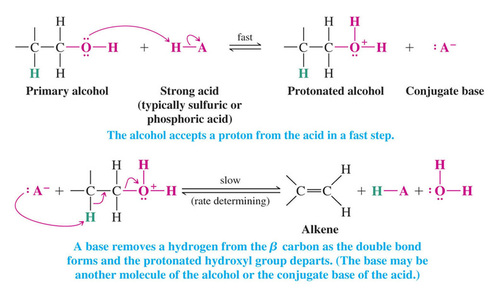
A Mechanism for Dehydration of Primary Alcohols: An E2 Reaction
>>Primary alcohols cannot undergo E1 dehydration because of the instability of the carbocation-like transition state in the 2nd step
>>In the E2 dehydration the first step is again protonation of the hydroxyl to yield the good leaving group water
>>The transition state for this reaction has carbocation character
>>Tertiary alcohols react the fastest because they have the most stable tertiary carbocation-like transition state in the second step
A Mechanism for Dehydration of Primary Alcohols: An E2 Reaction
>>Primary alcohols cannot undergo E1 dehydration because of the instability of the carbocation-like transition state in the 2nd step
>>In the E2 dehydration the first step is again protonation of the hydroxyl to yield the good leaving group water
Carbocation Stability and the Occurrence of Molecular Rearrangements
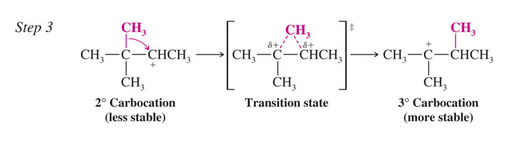
Rearrangements During Dehydration of Secondary Alcohols
Rearrangements of carbocations occur if a more stable carbocation can be obtained
Rearrangements During Dehydration of Secondary Alcohols
Rearrangements of carbocations occur if a more stable carbocation can be obtained
The first two steps are to same as for any E1 dehydration
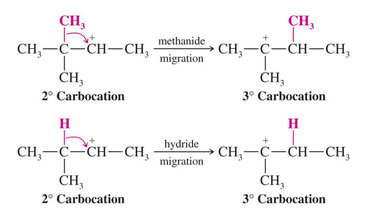
In the third step the less stable 2o carbocation rearranges by shift of a methyl group with its electrons (a methanide). This is called a 1,2 shift
The removal of a proton to form the alkene occurs to give the Zaitzev (most substituted) product as the major one
A hydride shift (migration of a hydrogen with its electrons) can also occur to yield the most stable carbocation
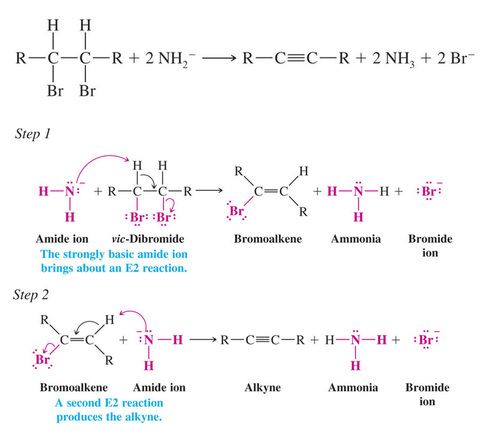
Synthesis of Alkynes by Elimination Reactions
Alkynes can be obtained by two consecutive dehydrohalogenation reactions of a vicinal dihalide.
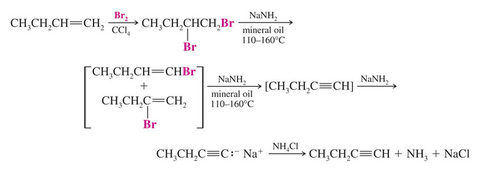
Alkenes can be converted to alkynes by bromination and two consecutive dehydrohalogenation reactions
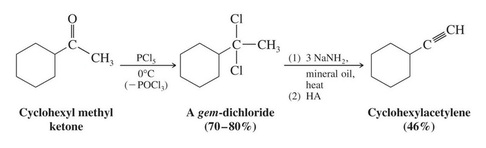
Geminal dihalides can also undergo consecutive dehydrohalogenation reactions to yield the alkyne
Download your lecture here!
Click on the links below to download the materials. All file formats are in Office 2007. Lectures were taken from Organic Chemistry by Solomons.
Chapter 1
Chapter 2
Alkanes and Cycloalkanes
Ionic Reactions - Substitution and Elimination
Alkenes and Alkynes: properties and Synthesis
Introduction to Organic reactions: Acids and Bases
Aromatic Compounds
Go to http://www.acdlabs.com/download/ to download Chemsketch.
ACD/ChemSketch is an advanced chemical drawing tool and is the accepted interface for the industry's best NMR and molecular property predictions, nomenclature, and analytical data handling software.
ACD/ChemSketch is an advanced chemical drawing tool and is the accepted interface for the industry's best NMR and molecular property predictions, nomenclature, and analytical data handling software.