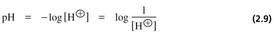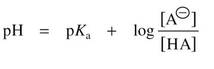Assignment...
|
AnnouncementsPlease print your experiment worksheet...
| ||||||||||||||||||||||||||
Lectures
|
| ||||||
| enzymes.docx | |
| File Size: | 166 kb |
| File Type: | docx |
| hormones.docx | |
| File Size: | 267 kb |
| File Type: | docx |
| vitamins.pptx | |
| File Size: | 1494 kb |
| File Type: | pptx |
| bloodbasics.pptx | |
| File Size: | 3609 kb |
| File Type: | pptx |
| dietary_macrominerals.pptx | |
| File Size: | 1798 kb |
| File Type: | pptx |
| dietary_microminerals.pptx | |
| File Size: | 1643 kb |
| File Type: | pptx |
NUCLEIC ACID
Download lecture HERE
PROTEIN
Download lecture HERE
LIPIDS
Download lecture HERE
CARBOHYDRATES
WATER
Principles of BIOCHEMISTRY, Third Edition (HORTON MORAN OCHS RAWN SCRIMGEOUR )
Water
• Water plays a central role in the chemistry of all life
• Proteins, polysaccharides, nucleic acids and membranes all assume their characteristic shapes in response to water
• The chemical properties of water are related to the functions of biomolecules, entire cells,and organisms
2.1 The Water Molecule is Polar
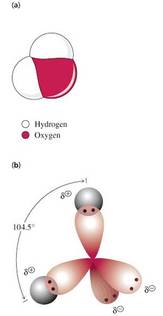
• Important properties of water arise from its angled shape
• Angle of 104.5o between two covalent bonds
• Polar O-H bonds due to uneven distribution of charge (oxygen (d-), hydrogen (d+))
• Angled arrangement of polar bonds creates a permanent dipole for a water molecule
• Water plays a central role in the chemistry of all life
• Proteins, polysaccharides, nucleic acids and membranes all assume their characteristic shapes in response to water
• The chemical properties of water are related to the functions of biomolecules, entire cells,and organisms
2.1 The Water Molecule is Polar
• Important properties of water arise from its angled shape
• Angle of 104.5o between two covalent bonds
• Polar O-H bonds due to uneven distribution of charge (oxygen (d-), hydrogen (d+))
• Angled arrangement of polar bonds creates a permanent dipole for a water molecule
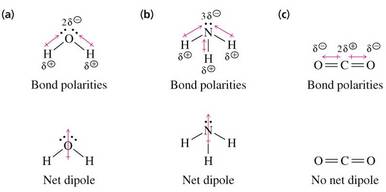
• Water and ammonia each have a permanent dipole while CO2 does not
2.2 Hydrogen Bonding in Water
• Water molecules attract each other due to their polarity
• A hydrogen bond is formed when a partially positive hydrogen atom attracts the partially negative oxygen atom of a second water molecule
• Hydrogen bonds can form between electronegative atoms and a hydrogen attached to another electronegative atom
• Water molecules attract each other due to their polarity
• A hydrogen bond is formed when a partially positive hydrogen atom attracts the partially negative oxygen atom of a second water molecule
• Hydrogen bonds can form between electronegative atoms and a hydrogen attached to another electronegative atom
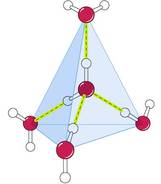
• A water molecule can form up to four hydrogen bonds
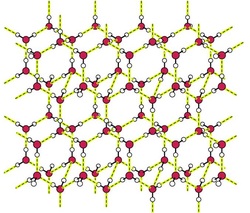
• Hydrogen bonds shown in yellow
• Hydrogen bonds shown in yellow
• Hexagonal lattice structure
• Every water molecule is H -bonded to 4 others
2.3 Ionic and Polar Substances Dissolve in Water
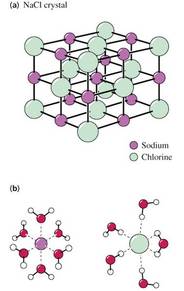
• Hydrophilic (water-loving) substances (polar and ionic (electrolytes)) readily dissolve in H2O
• Polar water molecules align themselves around ions or other polar molecules
• A molecule or ion surrounded by solvent molecules is solvated
• When the solvent is water the molecules or ions are hydrated
• Every water molecule is H -bonded to 4 others
2.3 Ionic and Polar Substances Dissolve in Water
• Hydrophilic (water-loving) substances (polar and ionic (electrolytes)) readily dissolve in H2O
• Polar water molecules align themselves around ions or other polar molecules
• A molecule or ion surrounded by solvent molecules is solvated
• When the solvent is water the molecules or ions are hydrated
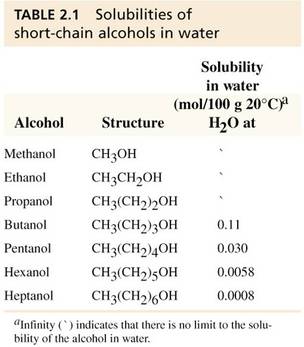
Solubilities of molecules in water
• Solubility in water depends upon the ratio of polar to nonpolar groups in a molecule
• The larger the portion of nonpolar groups the less soluble the molecule is in water
• The larger the portion of polar groups (e.g. hydroxyl groups (-OH)) the more soluble the molecule is in water
• Solubility in water depends upon the ratio of polar to nonpolar groups in a molecule
• The larger the portion of nonpolar groups the less soluble the molecule is in water
• The larger the portion of polar groups (e.g. hydroxyl groups (-OH)) the more soluble the molecule is in water
2.4 Nonpolar Substances Are Insoluble in Water
• Hydrophobic (water-fearing) molecules are nonpolar
• Hydrophobic effect - the exclusion of nonpolar substances by water (critical for protein folding and self-assembly of biological membranes)
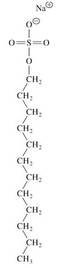
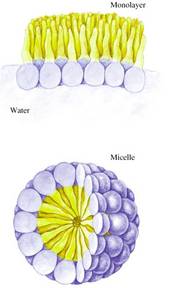
• Amphipathic molecules have hydrophobic chains and ionic or polar ends. Detergents (surfactants) are examples.
• Hydrophobic (water-fearing) molecules are nonpolar
• Hydrophobic effect - the exclusion of nonpolar substances by water (critical for protein folding and self-assembly of biological membranes)
• Amphipathic molecules have hydrophobic chains and ionic or polar ends. Detergents (surfactants) are examples.
2.5 Noncovalent Interactions in Biomolecules
Weak noncovalent interactions are important in:
• Stabilization of proteins and nucleic acids
• Recognition of one biopolymer by another
• Binding of reactants to enzymes
Noncovalent forces
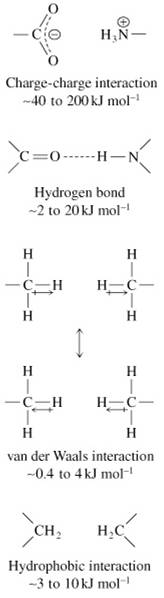
There are four major types of noncovalent forces:
(1) Charge-charge interactions
(2) Hydrogen bonds
(3) Van der Waals forces
(4) Hydrophobic interactions
A. Charge-Charge Interactions (Ion Pairing)
• Electrostatic interactions between two charged particles
• Can be the strongest type of noncovalent forces
• Can extend over greater distances than other forces
• Charge repulsion occurs between similarly charged groups
Types of attractive charged interactions
• Salt bridges - attractions between oppositely-charged functional groups in proteins
• Ion pairing - a salt bridge buried in the hydrophobic interior of a protein is stronger than one on the surface
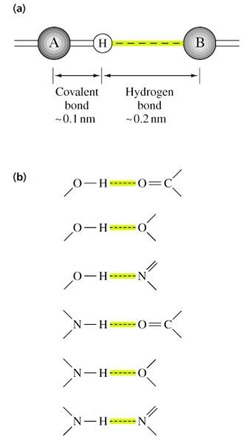
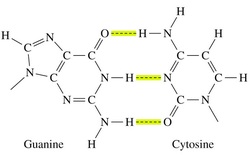
B. Hydrogen Bonds
• Among the strongest of noncovalent interactions
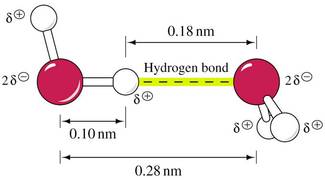
• H atom bonded to N, O, S can hydrogen bond to another electronegative atom (~0.2 nm distance)
• Total distance between the two electronegative atoms is ~0.27 to 0.30 nm
• In aqueous solution, water can H-bond to exposed functional groups on biological molecules
Weak noncovalent interactions are important in:
• Stabilization of proteins and nucleic acids
• Recognition of one biopolymer by another
• Binding of reactants to enzymes
Noncovalent forces
There are four major types of noncovalent forces:
(1) Charge-charge interactions
(2) Hydrogen bonds
(3) Van der Waals forces
(4) Hydrophobic interactions
A. Charge-Charge Interactions (Ion Pairing)
• Electrostatic interactions between two charged particles
• Can be the strongest type of noncovalent forces
• Can extend over greater distances than other forces
• Charge repulsion occurs between similarly charged groups
Types of attractive charged interactions
• Salt bridges - attractions between oppositely-charged functional groups in proteins
• Ion pairing - a salt bridge buried in the hydrophobic interior of a protein is stronger than one on the surface
B. Hydrogen Bonds
• Among the strongest of noncovalent interactions
• H atom bonded to N, O, S can hydrogen bond to another electronegative atom (~0.2 nm distance)
• Total distance between the two electronegative atoms is ~0.27 to 0.30 nm
• In aqueous solution, water can H-bond to exposed functional groups on biological molecules
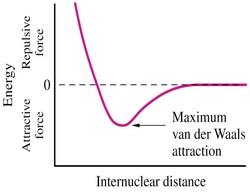
C. Van der Waals Forces
• Weak short range forces between:
(a) Permanent dipoles of two uncharged molecules
(b) Permanent dipole and an induced dipole in a neighboring molecule
• Although individually weak, many van der Waals interactions occur in biological macromolecules and participate in stabilizing molecular structures
• Weak short range forces between:
(a) Permanent dipoles of two uncharged molecules
(b) Permanent dipole and an induced dipole in a neighboring molecule
• Although individually weak, many van der Waals interactions occur in biological macromolecules and participate in stabilizing molecular structures
• Strongly repulsive at short internuclear distances, very weak at long internuclear distances
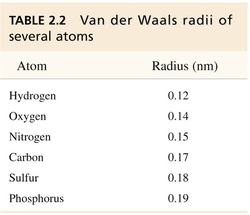
• Van der Waals attraction is maximal when two atoms are separated by their van der Waals radii
• Van der Waals attraction is maximal when two atoms are separated by their van der Waals radii
D. Hydrophobic Interactions
• Association of a relatively nonpolar molecule or group with other nonpolar molecules
• Depends upon the increased entropy (+DS) which occurs when water molecules surrounding a nonpolar molecule are freed to interact with each other in solution
• The cumulative effects of many hydrophobic interactions can have a significant effect on the stability of a macromolecule
• Association of a relatively nonpolar molecule or group with other nonpolar molecules
• Depends upon the increased entropy (+DS) which occurs when water molecules surrounding a nonpolar molecule are freed to interact with each other in solution
• The cumulative effects of many hydrophobic interactions can have a significant effect on the stability of a macromolecule
2.6 Water Is Nucleophilic
• Nucleophiles - electron-rich atoms or groups
• Electrophiles - electron-deficient atoms or groups
• Water is a relatively weak nucleophile
• Due to its high cellular concentration, hydrolysis reactions in water are thermodynamically favored
• Nucleophiles - electron-rich atoms or groups
• Electrophiles - electron-deficient atoms or groups
• Water is a relatively weak nucleophile
• Due to its high cellular concentration, hydrolysis reactions in water are thermodynamically favored
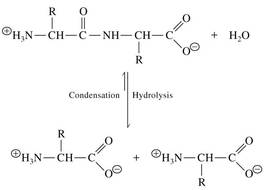
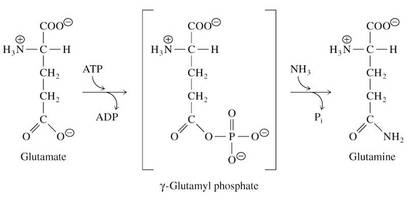
Condensation reactions can be favorable in cells
• ATP chemical energy can be used to drive reactions
• Glutamine synthetase catalyzes a condensation reaction
• ATP chemical energy can be used to drive reactions
• Glutamine synthetase catalyzes a condensation reaction
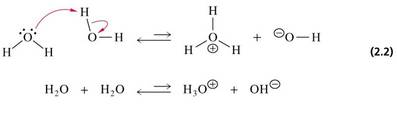
2.7 Ionization of Water
• Pure water consists of a low concentration of hydronium ions (H3O+) and an equal concentration of hydroxide ions (OH-)
• Acids are proton donors (e.g. H3O+) and bases are proton acceptors (e.g. OH-)
• Pure water consists of a low concentration of hydronium ions (H3O+) and an equal concentration of hydroxide ions (OH-)
• Acids are proton donors (e.g. H3O+) and bases are proton acceptors (e.g. OH-)
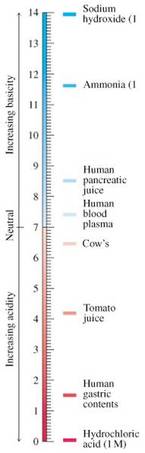
2.8 The pH Scale
• pH is defined as the negative logarithm of the concentration of H+
• pH is defined as the negative logarithm of the concentration of H+
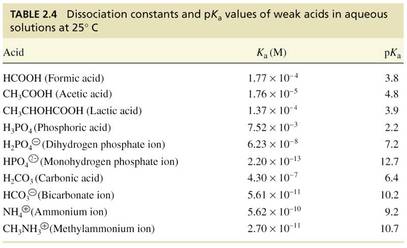
2.9 Acid Dissociation Constants of Weak Acids
• Strong acids and bases dissociate completely in water
HCl + H2O ßà Cl- + H3O+
• Cl- is the conjugate base of HCl
• H3O+ is the conjugate acid of H2O
Acetic acid is a weak acid
• Weak acids and bases do not dissociate completely in H2O
• Strong acids and bases dissociate completely in water
HCl + H2O ßà Cl- + H3O+
• Cl- is the conjugate base of HCl
• H3O+ is the conjugate acid of H2O
Acetic acid is a weak acid
• Weak acids and bases do not dissociate completely in H2O
The Henderson-Hasselbalch Equation
• Defines the pH of a solution in terms of:
(1) The pKa of the weak acid
(2) Concentrations of the weak acid (HA) and conjugate base (A-)
• Defines the pH of a solution in terms of:
(1) The pKa of the weak acid
(2) Concentrations of the weak acid (HA) and conjugate base (A-)
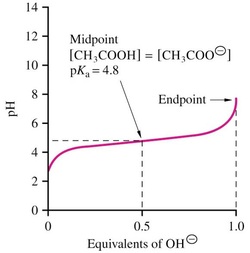
2.10 Buffered Solutions Resist Changes in pH
• Buffer capacity is the ability of a solution to resist changes in pH
• Most effective buffering occurs where:
solution pH = buffer pKa
• At this point: [weak acid] = [conjugate base]
• Effective buffering range is usually at pH values equal to the pKa ± 1 pH unit
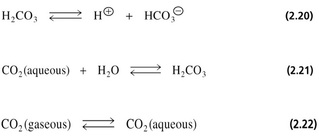
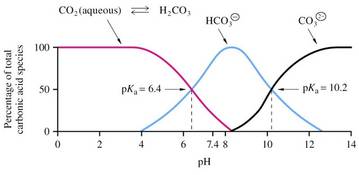
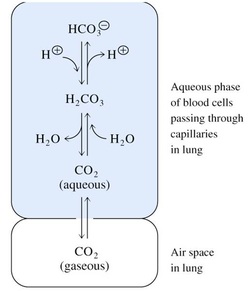
Regulation of pH in the blood of animals
• Blood plasma of mammals has a constant pH which is regulated by a buffer system of:
carbon dioxide /carbonic acid /bicarbonate
• Buffer capacity depends upon equilibria between:
(1) Gaseous CO2 (air spaces of the lungs)
(2) Aqueous CO2 (dissolved in the blood)
(3) Carbonic acid
(4) Bicarbonate
• Buffer capacity is the ability of a solution to resist changes in pH
• Most effective buffering occurs where:
solution pH = buffer pKa
• At this point: [weak acid] = [conjugate base]
• Effective buffering range is usually at pH values equal to the pKa ± 1 pH unit
Regulation of pH in the blood of animals
• Blood plasma of mammals has a constant pH which is regulated by a buffer system of:
carbon dioxide /carbonic acid /bicarbonate
• Buffer capacity depends upon equilibria between:
(1) Gaseous CO2 (air spaces of the lungs)
(2) Aqueous CO2 (dissolved in the blood)
(3) Carbonic acid
(4) Bicarbonate